A pharma plant can lose hours when one phone fails after a washdown, or when a wrong Ex rating blocks commissioning. Both problems are avoidable.
Select the right explosion-proof telephone by matching the site’s Zone/Division and gas/dust group first, then demand hygienic design for GMP cleaning, and finally prove SIP emergency call routing with a complete audit-ready document pack.

A practical selection method that survives GMP audits and real washdowns?
A pharmaceutical workshop has two strong forces. One force is safety, because solvents and powders can create explosive atmospheres. The other force is hygiene, because GMP 1 cleaning and disinfectants attack materials and seals. A phone that is “ATEX approved” can still fail hygiene, and a phone that is “stainless and washable” can still fail Ex needs. The selection process has to join both.
The most reliable method uses four gates:
Gate 1: Hazard fit (where it can be installed)
The zone and group rating must match the exact mounting point. In practice, solvent transfer points and powder discharge points often need stricter ratings than nearby walkways. A single “one model fits all” choice can waste budget, but it also reduces mistakes when maintenance swaps devices. Both approaches can work, but the decision should be planned.
Gate 2: Hygiene fit (how it can be cleaned)
Cleaning crews will use alcohol, peroxide, quats, or mixed chemistry. If the keypad swells, or if the gasket hardens, the phone starts leaking. That causes corrosion and downtime. A hygienic phone needs smooth surfaces, sealed interfaces, and chemical-resistant elastomers.
Gate 3: Emergency fit (how it will be used)
Pharma sites often need direct calling to EHS, security, and a control room. A phone must support hotline calls, priority routing, and clear location identity. It should also provide remote checks so IT can find failures early.
Gate 4: Evidence fit (how it will pass audits)
A buyer needs proof that the model shipped is the model certified and controlled. That includes certificates, declarations, production QA approvals, test reports, and a spare-parts plan that prevents “unapproved substitutions.”
| Gate | Main question | What must be true | Output of the gate |
|---|---|---|---|
| Hazard fit | Is this point classified? | Rating matches Zone/Division and group | Approved Ex marking for each location |
| Hygiene fit | Can it survive cleaning? | Smooth design + compatible materials | Cleaning and material compatibility plan |
| Emergency fit | Will calls reach the right people? | SIP routing and power resilience work | Tested call flows and failover |
| Evidence fit | Can it pass audits? | Docs match, QA is valid, spares are controlled | Audit-ready supplier approval pack |
A short story helps explain why this order matters. On one project, the team bought a strong Zone phone first and ignored disinfectant exposure. The keypad then cracked during routine cleaning. The Ex rating did not save uptime. The fix was a hygienic membrane keypad and better elastomer selection.
This selection method keeps the phone safe, cleanable, and usable during real incidents.
Now the process needs concrete answers for four topics: classification and groups, hygienic design, SIP and PoE planning, and the document pack for audits.
Which hazardous-area classification (Zone/Division) and gas or dust group should be specified for solvent handling and powder processing areas?
Wrong classification creates a hidden project risk. The phone may be installed, but it can be rejected later, or it can be unsafe at the worst time.
Use the site’s hazardous-area study to set Zone/Division per mounting point, then specify gas group for solvent vapors and dust group for powders. If both hazards exist, require dual gas + dust certification.
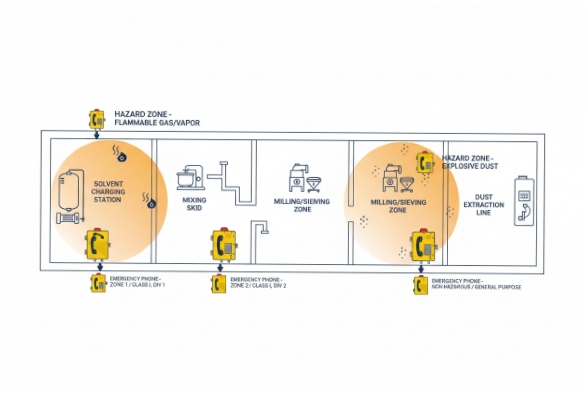
Use site data, not “typical rules”
A pharma workshop can include solvent charging, blending, filling, coating, milling, sieving, and packaging. Each step changes the likelihood of release. A phone point near an open solvent drum station is not the same as a phone point on a corridor wall. The correct input is the plant’s classification drawings and risk study. If those are missing, the safest path is to treat the area as potentially higher risk until the owner confirms it.
Solvent handling: gas/vapor classification basics
Solvent areas are often classified as Zone 1 2 or Zone 2 in IEC/ATEX style, or Division 1 or Division 2 in NEC style. The classification depends on how often vapor can be present. Transfer points, vent points, and sampling points often drive stricter zones. Gas group selection depends on the solvent type and the worst credible vapor. Many buyers specify a higher gas group to cover more cases, but the phone must still match temperature limits and ambient range.
Powder processing: dust classification basics
Powder processing can create dust clouds during charging, discharge, sieving, and vacuum transfer. In IEC/ATEX style, this often maps to Zone 21 or Zone 22. Dust group depends on dust conductivity and particle behavior. Many pharma powders are organic and non-conductive, but some processes involve conductive dust or mixed powders. The safest approach is to use dust explosivity data when it exists, and to avoid assumptions.
Mixed hazards: demand a clear answer
Some rooms have both solvents and powders. That can happen in coating, granulation, or cleaning steps. In those cases, a phone should be selected with gas and dust marking that matches both. The specification should also state if the device must keep its rating with the chosen cable entry method.
| Area in a pharma workshop | Main hazard | Likely classification pattern (site-dependent) | What to specify for a phone point |
|---|---|---|---|
| Solvent drum charging / mixing skid | Vapor release | Often Zone 1 or Div 1 near the source | Gas-certified model, correct gas group, safe surface temp |
| Solvent storage room corridor | Vapor migration | Often Zone 2 or Div 2 | Gas-certified model for Zone 2/Div 2, strong sealing |
| Milling / sieving / bag dumping | Dust cloud | Often Zone 21 or Div 1 (dust) close to process | Dust-certified model, correct dust group, washdown sealing |
| Packaging with dust capture | Residual dust | Often Zone 22 or Div 2 (dust) | Dust-certified model, simpler category allowed if approved |
| Coating or hybrid room | Vapor + dust possible | Mixed zones in one area | Dual gas + dust certified model with clear marking |
A good RFQ does not ask for “an ATEX phone.” It lists each mounting point, the classification reference, and the required marking. That single step prevents most selection errors.
What hygienic design features—stainless housing, smooth surfaces, sealed keypad, and washdown IP rating—support GMP cleaning and disinfectants?
Cleaning is not gentle. It uses pressure, heat cycles, and chemicals. A phone must keep its seals, finish, and readability through years of cleaning.
Choose hygienic design with 316L stainless housing, smooth and drainable geometry, sealed membrane keypad, and a washdown-ready IP rating. Confirm gasket and keypad materials resist the site’s disinfectants and cleaning cycles.

Stainless housing and surface finish that cleaning teams can trust
Stainless steel is popular in pharma because it resists corrosion and looks clean. For harsh cleaning, 316L 3 is often preferred. Smooth finish matters as much as the grade. Rough surfaces hold residues and slow cleaning. A phone enclosure should have smooth planes, rounded edges, and minimal gaps. Labels should be durable and easy to wipe without peeling.
A good hygienic design also avoids “dirt traps.” That includes deep recesses around keypads, sharp seams, exposed springs, and uncovered screw heads where residue can build. If fasteners are needed, they should be corrosion resistant and easy to clean.
Sealed keypad and audio openings that resist liquids
Keypads are a frequent failure point in washdown areas. A sealed membrane keypad reduces ingress and makes wiping easier. The keypad should not bubble, crack, or swell under disinfectants. The microphone and speaker openings need protection that blocks water and still supports clear audio. A protective membrane can help, but it must be part of the rated design, not an aftermarket addition.
IP rating: pick it based on the real washdown method
Many pharma plants use wet cleaning, foam cleaning, or wipe-down. Some lines use higher pressure sprays. IP66 is often a baseline for strong jets. IP67 helps when temporary immersion or pooling water can occur. In some washdown regimes, an even stronger washdown performance may be required. The key is to match the plant’s cleaning SOP, not to chase the highest number.
Chemical compatibility: make it a written matrix
Disinfectants vary. IPA and ethanol can dry out some elastomers. Peroxide and peracetic acid can attack others. Quats can leave films. A supplier should confirm gasket, keypad, and handset material compatibility. This is best handled with a simple matrix tied to the site chemicals.
| Hygienic feature | Why it matters | What to verify during evaluation | Typical failure if ignored |
|---|---|---|---|
| 316L stainless housing | Resists corrosion and frequent wipe | Material grade evidence and external hardware grade | Pitting and rust at screws and hinges |
| Smooth, rounded geometry | Reduces residue and speeds cleaning | No deep seams, drain-friendly shape | Residue build-up and cleaning non-compliance |
| Sealed membrane keypad | Prevents ingress and makes wiping easy | Chemical resistance and life-cycle presses | Swelling, cracking, stuck keys |
| Washdown IP rating | Protects internals from jets and splashes | IP test evidence and cable entry method | Water ingress at glands and audio openings |
| Chemical-resistant gaskets | Keeps IP and hygiene over time | Compatibility with disinfectants used on site | Gasket hardening and slow leaks |
A hygienic Ex phone is a system. The enclosure, keypad, glands, and labels must all survive cleaning. A supplier that only talks about Ex marking but cannot talk about disinfectants is not ready for pharma workshops.
How should SIP/VoIP integration, PoE power, and emergency call routing be planned for pharma plants using IP PBX, dispatch, and access control?
A phone that “registers” is not yet an emergency phone. Emergency routing needs priority, location, and power resilience. Pharma sites also need controlled change and stable uptime.
Plan SIP integration by testing real call flows on the target IP PBX and dispatch system, using PoE with UPS-backed switches, and setting emergency routes with hotline, priority, and location identity. Link access control actions only when they are approved and tested.

Network and PoE planning: make power and VLAN part of the design
PoE is convenient, but it depends on switch health and cable quality. In pharma plants, switches often sit in controlled rooms, while phones sit in washdown areas. The design should include:
-
Redundant uplinks where the plant needs high uptime
-
Voice VLAN and QoS markings so calls stay clear during traffic spikes
-
A clear rule for where SIP signaling and RTP media are allowed through firewalls
A phone should also recover cleanly after power loss. Boot time matters. A long boot can feel like a failure during an incident.
Emergency call routing: build it around roles and locations
Emergency calls should reach the right role fast. Common targets include EHS, security, the control room, and maintenance. Routing can use hotline keys, speed dials, or auto-dial on off-hook. The design should also ensure that the dispatcher sees location details. A simple location naming standard helps, like “WS-03 Solvent Room North Door.”
Dispatch integration should be tested with group calling, call recording rules, and callback behavior. If the site uses paging or broadcasting, the phone may also need to trigger paging zones or speak into a PAGA 5 path through gateways or SIP paging features.
Access control integration: keep it intentional
Some pharma plants want a phone to trigger a door release or a gate action. This can be useful, but it also creates risk if it is not controlled. If access control integration is needed, the system should define:
-
who can trigger it
-
which events trigger it
-
how it is logged
-
how it fails safe
Testing plan: short, repeatable, and close to real life
A good acceptance test is simple:
1) Hotline call to dispatch
2) Verify caller ID and location label
3) Priority call during a busy condition
4) Power cycle PoE and confirm auto-recovery
5) Paging or alert test if needed
| Integration item | What to check | Pass criteria for emergency calling | Common miss |
|---|---|---|---|
| SIP PBX registration | Auth, keepalive, failover | Stable registration and fast recovery | No test after power loss |
| Hotline and priority | Busy override and routing rules | Call reaches emergency group every time | Hotline works only in normal load |
| Dispatch console | Identity and callback | Clear location and easy callback | Location naming is inconsistent |
| PoE and UPS | Resilience | Phone stays available during power dips | Switch not on UPS |
| Access control link | Security and logging | Controlled trigger and full logs | Door release works but is not audited |
Pharma plants also care about controlled changes. Firmware updates and configuration changes should follow a site change process. A phone that supports remote configuration backup and clear logs makes that process easier and reduces downtime.
Which compliance documents should be verified—ATEX/IECEx, EU Declaration of Conformity, QAN/QAR, test reports, and spare parts—to pass project audits?
A project audit often fails for one simple reason: the documents do not match the delivered model, or the factory control is unclear. Fixing that late is expensive.
Verify that certificates, declarations, and factory QA approvals match the exact model and configuration. Request IP and corrosion evidence, installation instructions, change-control policy, and a controlled spare-parts list tied to certified revisions.

Certificates and declarations: match the nameplate, not the marketing sheet
The phone label must match the certificate marking and the EU Declaration of Conformity 6 for EU projects. Buyers should check model codes, options, ambient limits, and any special conditions. Certificate schedules often list allowed cable entries and glands. If the plant uses a different gland, the compliance chain can break.
IECEx documents help when the project spans regions or uses global standards. ATEX documents matter for EU installations. Both can exist for the same product family, but the buyer must confirm that the exact ordered variant is covered.
Production QA approvals: confirm ongoing control, not one-time testing
A compliant Ex phone also needs controlled production. This is where QAN/QAR style approvals matter. The audit should confirm:
-
the factory name and address
-
the approval scope that covers the protection concept
-
the validity and surveillance status
-
traceability from serial number to production batch
Test reports: ask for proof tied to pharma reality
Pharma teams often ask for more than Ex compliance. They need washdown and chemical resistance confidence. Useful evidence includes:
-
IP test reports
-
corrosion or salt spray evidence for stainless and hardware
-
material statements for enclosure and fasteners
-
keypad and gasket chemical resistance notes
-
functional test evidence for hotline and emergency calling
Spares and maintenance: lock down what can be replaced
A spare part plan reduces downtime, but it must not break compliance. A supplier should provide a spare list with part numbers, revision control, and service rules. If a keypad or handset is replaced, it must be the approved part. The same is true for cable glands and entry accessories.
| Document or item | What it proves | What to verify | Decision rule |
|---|---|---|---|
| ATEX/IECEx certificates + schedules | Ex suitability and conditions | Marking, options, Ta range, entries | Reject if schedules do not match configuration |
| EU Declaration of Conformity | Legal conformity for EU market | Correct entity, model, standards list | Reject if model name differs from label |
| QAN/QAR or production QA evidence | Factory audits and ongoing control | Factory address, scope, validity | Reject if factory differs or scope is unclear |
| IP / washdown evidence | Suitability for GMP cleaning | Test method, rating, entry method | Reject if no evidence for cleaning regime |
| Installation and safety instructions | Correct mounting and cable entry | Gland rules, torque, warnings | Reject if entry rules are vague |
| Spare parts kit + BOM control | Fast repair without compliance risk | Part numbers, approved substitutions | Reject if spares are “generic” |
| Firmware/change-control policy | Ongoing compliance and stability | Versioning, change impact review | Reject if changes are uncontrolled |
An audit-ready supplier is the one that treats documents and change control as part of the product. That supplier can support validation packs, factory traceability, and stable spares for years.
Conclusion
Match classification first, then demand hygienic washdown design, prove SIP emergency routing under real conditions, and approve suppliers only with complete certificates, QA control, and controlled spares.
Footnotes
-
Good Manufacturing Practice regulations ensuring products are consistently produced and controlled. ↩
-
Hazardous area classification where explosive gas atmosphere is likely to occur in normal operation. ↩
-
Low-carbon austenitic stainless steel highly resistant to corrosion and commonly used in cleanrooms. ↩
-
Uninterruptible Power Supply providing emergency power when the input source fails. ↩
-
Public Address and General Alarm system used for site-wide broadcasting and alerts. ↩
-
Official document stating that a product meets all relevant requirements of applicable EU directives. ↩











